JOURNAL 3780
Organic Communications
VOLUME & ISSUE
Year: 2026 Issue: 1
Year: 2026 Issue: 1
PAGES
p.2 - 2
p.2 - 2
STATISTICS
Viewed 370 times.
Viewed 370 times.
GRAPHICAL ABSTRACT
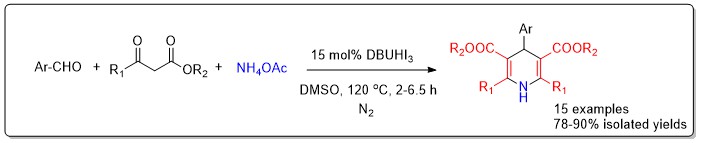
ABSTRACT
A process-optimized, one-pot multicomponent reaction catalyzed by DBUHI3 was developed, enabling the synthesis of diverse 1,4-dihydropyridines (1,4-DHPs), a class of bioactive nitrogen heterocycles from substituted benzaldehydes, ethyl acetoacetate, and ammonium acetate under mild conditions in DMSO. The methodology afforded good to excellent yields for a variety of aryl aldehyde substrates bearing both electron-donating and electron-withdrawing groups. Structural confirmation was achieved through IR, 1H NMR, 13C NMR, and HRMS analyses. This work not only summarizes key synthetic strategies but also provides a practical, sustainable route to the preparation of 1,4-DHPs, supporting further medicinal and synthetic exploration.
KEYWORDS- 1,4-dihydropyridines (1,4-DHPs)
- DBUHI3
- organocatalyst
- multicomponent reaction (MCR)