Journal of Chemical Metrology
A scientific open access journal in the field of analytical chemistry and accreditationLATEST ARTICLES
Metrological framework for the development of plant-based certified referencematerials. Part I
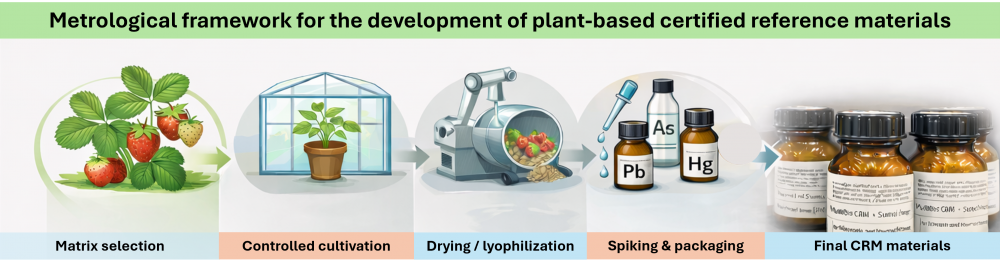
Certified reference materials constitute a fundamental principle of reliable measurements across scientific, industrial, and regulatory domains, as they provide the basis for the formal demonstration of metrological traceability and result comparability. Unfortunately, the availability of certified reference materials for complex plant matrices, particularly fruit or leaves tissues, remains limited. This study presents the scenario for the development of MultiBio CRM, a strawberry-based certified reference material pair comprising leaves and fruit, which reflects compositional characteristics typical of plant materials cultivated under Polish environmental and agricultural conditions. The study further demonstrates a complete, ISO-aligned workflow for the production of plant-based reference materials. The production strategy was designed in accordance with ISO 17034, ISO 33405, and related international requirements for reference material production. It encompasses matrix selection, controlled cultivation and sampling, pre-treatment, drying or lyophilization, particle-size reduction, homogenization, stabilization, and certification. Material homogeneity and stability were verified using statistically designed studies, and uncertainty contributions were evaluated following internationally accepted guidelines. The resulting certified reference materials represent robust, internationally compliant plant matrices and illustrate a comprehensive metrological approach to reference material development. This work addresses a significant gap in existing certified reference material portfolios and supports reliable, traceable measurements in food-safety and environmental analysis.
DOI http://doi.org/10.25135/jcm.2602.3803 Keywords Certified reference materials plant-based reference materials reference material production ISO 17034 ISO 33405 metrological traceability DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.Desorption of ethanol from internal surface of the cylinders

Reference materials (RMs) are essential for ensuring metrological traceability in various fields, including environmental monitoring, chemical analysis, and the verification of alcohol breath analyzers. One critical property of RMs is their long-term stability, particularly for gas mixtures such as ethanol in nitrogen, which are stored in pressurized aluminum cylinders. This paper describes the preparation of such gas mixtures according to ISO 6142-1, utilizing differential weighing to minimize the influence of ambient conditions and ensure accurate composition. The effects of adsorption of ethanol on the internal surfaces of aluminum cylinders were experimentally investigated. Results indicate that ethanol can remain adsorbed on the cylinder walls even after evacuation, leading to measurable ethanol concentrations upon subsequent refilling with nitrogen. Repeated fillings demonstrated a decrease in ethanol levels, confirming the saturation and gradual desorption behavior. These findings underscore the necessity to consider adsorption effects in the uncertainty budget and composition calculations for low-concentration ethanol-in-nitrogen RMs.
DOI http://doi.org/10.25135/jcm.121.2507.3596 Keywords Stability reference materials ethanol pressurized cylinders gas chromatography desorption DETAILS PDF OF ARTICLE © 2025 ACG Publications. All rights reserved.Development of a quality control material for conductivity measurements in food and environmental applications
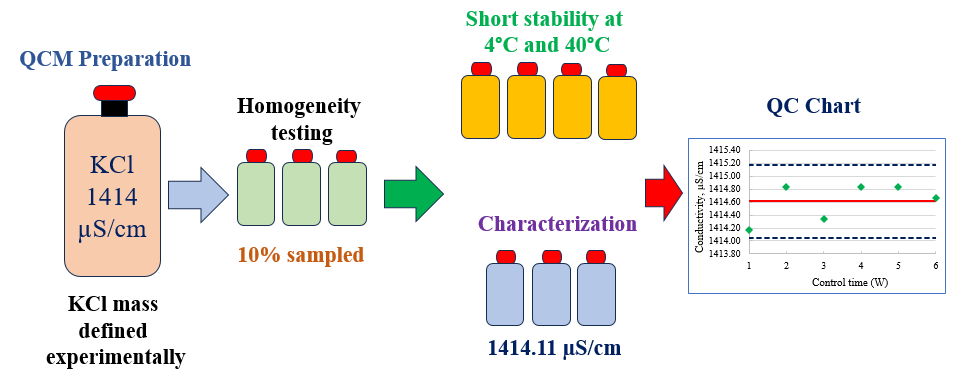
The accuracy and reliability of conductivity measurements in analytical laboratories depend significantly on the availability of quality control materials. This study addresses the preparation, homogeneity, stability and characterization of a potassium chloride (KCl) quality control material (QCM) of 1414 µS/cm based on ISO/TR 33402. OIML R-56 does not contain this conductivity value as a secondary standard, so the mass of KCl required to prepare such a solution was experimentally defined. The conductivity measurements were carried out at 25 °C using a conductivity meter calibrated by a CRM produced by the the Slovak Institute of Metrology (SMU), a signatory to the mutual recognition arrangement (MRA) of the International Committee of Weights and Measures (CIPM). The homogeneity study was carried out in accordance with ISO 33405 using 10% of the batch bottles and the analysis of variance (ANOVA) showed that the QCM batch is homogeneous. The short-term stability was carried out over 4 weeks storage time at 4°C and 40°C and the isochronous measurements showed no significant deviations over time. The characterization of the QCM along three days showed that, its conductivity was 1414.11 µS/cm. The uncertainty associated with the conductivity measurements was assessed based on the requirements of the Guide to the expression of uncertainty in measurement (ISO GUM) and the EURACHEM/CITAC Guide, CG4 (Quantifying uncertainty in analytical measurement). It was found to be 23.10 µS/cm or 1.63%. A control chart was developed using the prepared QCM and the measured values remained within the control limits over the control time of six weeks. The prepared KCl QCM will be useful for use in quality control and instrumental validation in food, drug and environmental conductivity testing.
DOI http://doi.org/10.25135/jcm.119.2508.3607 Keywords Conductivity QCM ISO/TR 33402 homogeneity stability control chart DETAILS PDF OF ARTICLE © 2025 ACG Publications. All rights reserved.Determination of phenolic content of Hypericum aucheri Jaub. & Spach by LC-HRMS and its antioxidant capacity

The study aims to identify the secondary metabolites of Hypericum aucheri Jaub. & Spach extracts using liquid chromatography-high resolution mass spectrometry (LC-HRMS) and to evaluate the plant's antioxidant activity. Antioxidant activity was assessed using DPPH free radical scavenging and CUPRAC assays. The primary secondary metabolite detected was hyperoside, with concentrations of 1059.53 mg/L in acetone (Ac) extract and 389.73 mg/L in methanol (MeOH) extract. Sinapinic acid was the predominant compound in the chloroform (C) extract, with a concentration of 6646.53 mg/L. In addition, Ac extract showed the highest DPPH free radical scavenging activity (85.90±0.26 - 61.19±0.47%) of all the extracts tested. The MeOH extract exhibited the highest value in the CUPRAC assay (2.53±0.05 mmol TR g-1). The study highlights a numerical relationship between phenolic content and antioxidant activity, underscoring the importance of phenolics in their antioxidant functions.
DOI http://doi.org/10.25135/jcm.131.2512.3765 Keywords Hypericaceae Hypericum aucheri antioxidant LC-HRMS phenolic compounds DETAILS PDF OF ARTICLE © 2025 ACG Publications. All rights reserved.