Records of Natural Products
Year: 2026 Volume: 20 Issue:4
1) A mixture of potent polyphenolic anticarcinogens: Microarray analysis of their efficacy in MCF-7 and MCF-10A cell lines
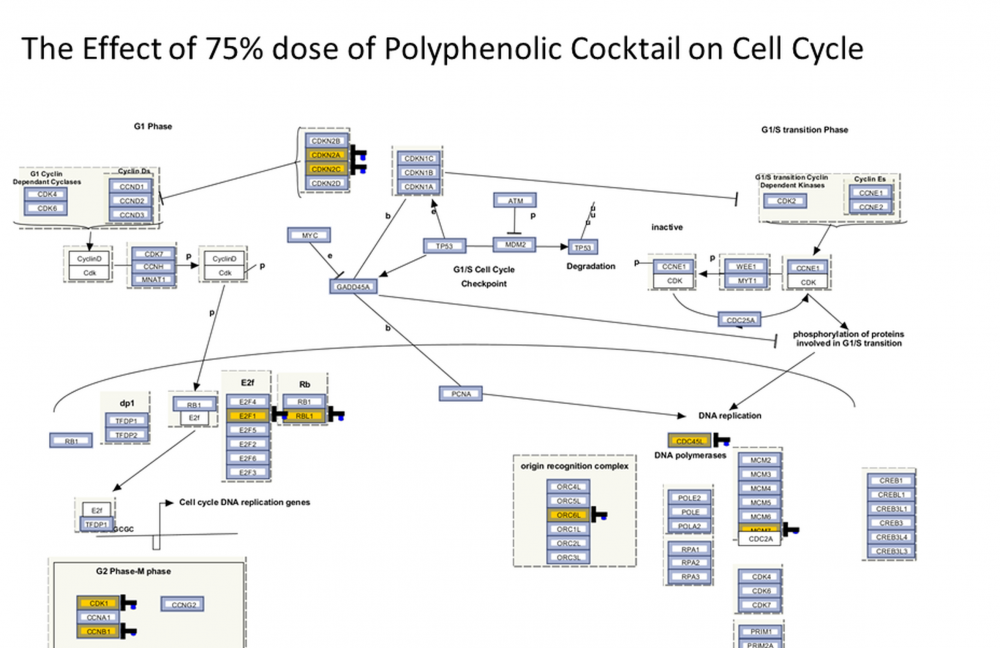
Polyphenols are abundantly available through diet and show great promise with their safe use against cancer. In our previous studies, we have shown cytotoxic effects of more than 30 polyphenols in breast cancer. In this study, we have aimed to investigate the effects of various polyphenols upon MCF-7 breast cancer and MCF-10A normal epithelial cells. To this end, we have designed a polyphenolic mixture based on the most effective concentrations of seven compounds, which is a first according to literature in terms of polyphenolic combination studies. Cell proliferation experiments were executed utilizing WST-1 and apoptotic status by Annexin V-PI. After determining most effective concentrations, we operated whole genome microarray analysis. Based on microarray data, the best effective concentration for both cell lines is 75% of polyphenolic mixture. In MCF-7 cell line, this dosage induced in down regulation of cell cycle, cell division, DNA repair and some genes linked to breast cancer. In contrast, no remarkable effect was observed in MCF-10A cell line. The designed polyphenolic mixture was demonstrated to inhibit breast cell division in multiple pathways. Our findings on cell division in MCF-7 cell line were found similarly with the study based on polyphenol-rich propolis which inhibited cell division remarkably in SK-BR-3 cells. This mixture, which is shown to have highly effective anticarcinogenic effects, can be considered as a prototype of natural prescription design for our future research.
DOI http://doi.org/10.25135/rnp.2510.3658 Keywords Polyphenols breast cancer whole genome expression cell proliferation cell division microarray DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.2) Cyclocarioside Z15, a new 3,4-seco dammarane triterpenoid glycoside fromthe leaves of Cyclocarya paliurus with α-glucosidase inhibitory activity
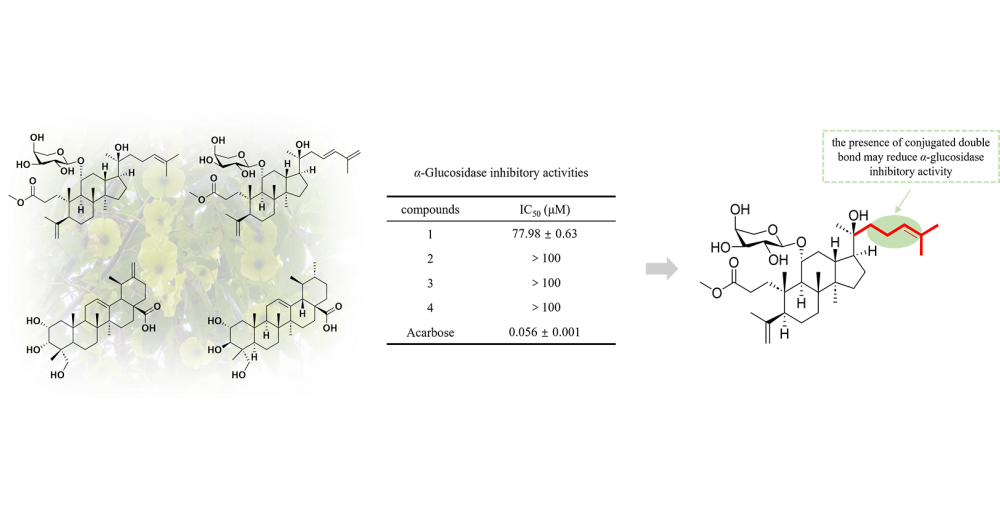
A new 3,4-seco dammarane triterpenoid glycoside, cyclocarioside Z15 (1), along with three known compounds cypaliuruside B (2), 2α,3α,23-trihydroxyurs-12,20(30)-dien-28-oic acid (3) and asiatic acid (4) were isolated from the leaves of Cyclocarya paliurus. The chemical structures were elucidated by 1D and 2D NMR, HR-ESI-MS, and acid hydrolysis. All compounds were measured for their α-glucosidase inhibitory activity, and only compound 1 exhibited weak activity with an IC50 value of 77.98 μM.
DOI http://doi.org/10.25135/rnp.2602.3800 Keywords Cyclocarya paliurus 3,4-seco dammarane triterpenoid glycoside α-glucosidase inhibitory activity DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.3) Inhibition of the NLRP3 inflammasome by Callinudin A: A novel 3,4-seco-labdane diterpenoid derived from Callicarpa nudiflora
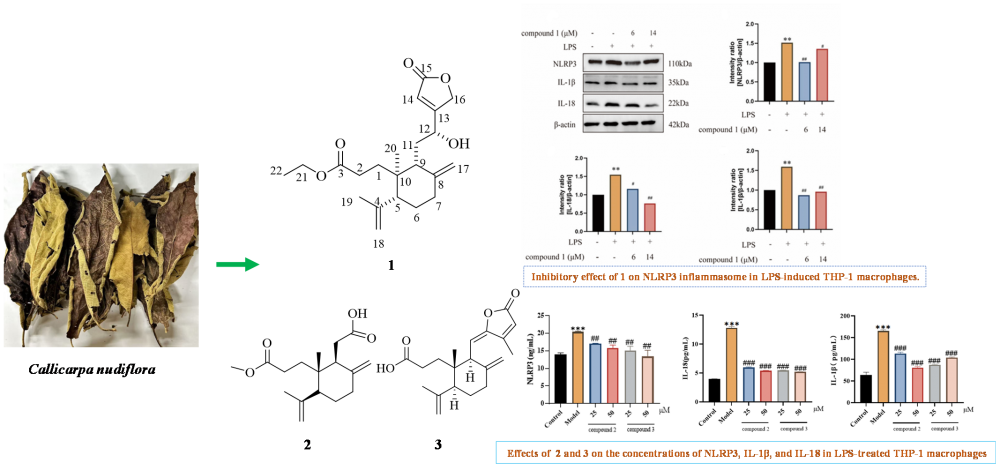
A previously uncharacterized 3,4-seco-labdane diterpenoid, which is designated Callinudin A (1), along with two known diterpenoids of the same type (2, 3), was isolated from Callicarpa nudiflora leaves. Multiple spectroscopic techniques and literature comparisons were used to elucidate the structures of these compounds. Furthermore, these compounds were tested in vitro to assess their effects on the NLRP3 inflammasome in lipopolysaccharide (LPS)-induced THP-1 macrophages. Analysis revealed that these compounds exhibit inhibitory effects on the NLRP3 inflammasome.
DOI http://doi.org/10.25135/rnp.2512.3770 Keywords Callicarpa nudiflora Callinudin A 3,4-seco-labdane diterpenoid NLRP3 inflammasome DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.4) Germicidin T: A new trimethyl-substituted α-pyranone fromthe Mangrove-derived Streptomyces ardesiacus GXIMD 03502
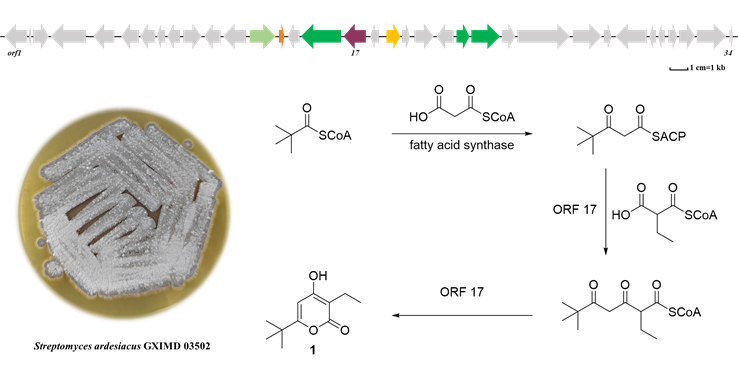
An in-depth investigation into the chemical constituents of Streptomyces ardesiacus GXIMD 03502, a strain known to produce 2-hydroxyphenylthiazoline derivatives, resulted in the isolation of a new α-pyrone compound, germicidin T (1), together with six known compounds (2–7). Their structures were elucidated using comprehensive spectroscopic analyses. The germicidin biosynthetic gene cluster was identified in the genome of this strain. Notably, ORF 17, a type III polyketide synthase (Germicidin synthase, GCS), was found to utilize trimethylmalonyl-ACP as a starter unit in the biosynthesis of compound 1. Based on this finding, the biosynthetic pathway for compounds 1–7 is proposed. Bioactivity evaluation revealed that all compounds exhibited weak toxicity against Artemia salina, while compounds 3 and 7 showed moderate anti-inflammatory activity.
DOI http://doi.org/10.25135/rnp.2601.3792 Keywords Streptomyces ardesiacus α-pyrone germicidin trimethylmalonyl-ACP type III polyketide synthase biosynthesis DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.5) Bioactive compounds from the cultured lichenmycobiont of Nigrovothelium inspersotropicum
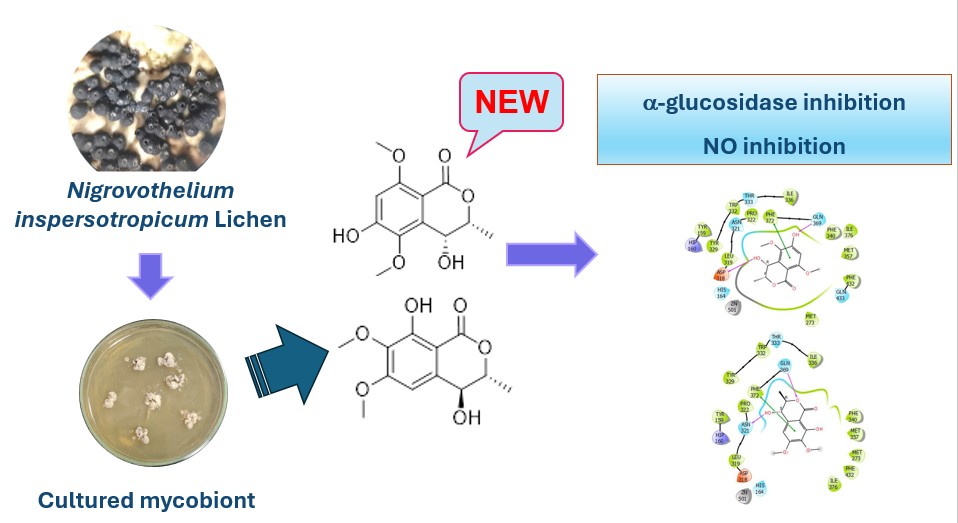
Cultured lichen mycobionts have been considered as valuable sources of natural compounds with unique structure scaffolds. Nigrovothelium inspersotropicum, a crustose lichen from the family Trypetheliaceae, is an indigenous species in Vietnam. However, there is a scarcity of chemical and biological information available regarding N. inspersotropicum and its cultured mycobionts. In this study, the mycobiont of N. inspersotropicum was cultivated and subjected to chemical and biological investigations. As a result, six compounds, including 3,4-dihydro-7,8-dihydroxy-6-methoxy-3-methylisocoumarin (1), (+)-(3S)-6,7-dimethoxymellein (2), 8-hydroxy-6,7-dimethoxyisocoumarin (3), aspermytin A (4), subnudatone B (5), and b-sitosterol (6), were isolated and structurally elucidated using extensive spectroscopic analyses (1D- and 2D-NMR and HRESIMS). Notably, compound 1 is new compound, while compound 2 is identified as a new natural compound. The isolated compounds were evaluated for their inhibitory activities against alpha-glucosidase, nitric oxide production, SARS-CoV-2 Mpro, and HIV-1 reverse transcriptase. In summary, this study presents new information on natural compounds derived from the mycobiont of N. inspersotropicum and their promising anti-inflammatory, antihyperglycemic, and antiviral activities.
DOI http://doi.org/10.25135/rnp.2511.3727 Keywords Nigrovothelium inspersotropicum isocoumarin alpha-glucosidase inhibition NO inhibition HIV-1 reverse transcriptase DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.6) Unveiling the phytochemical landscape of Phlomis pungens willd.: LC-MS/MS insights into antioxidant and multi-enzyme inhibitory potential
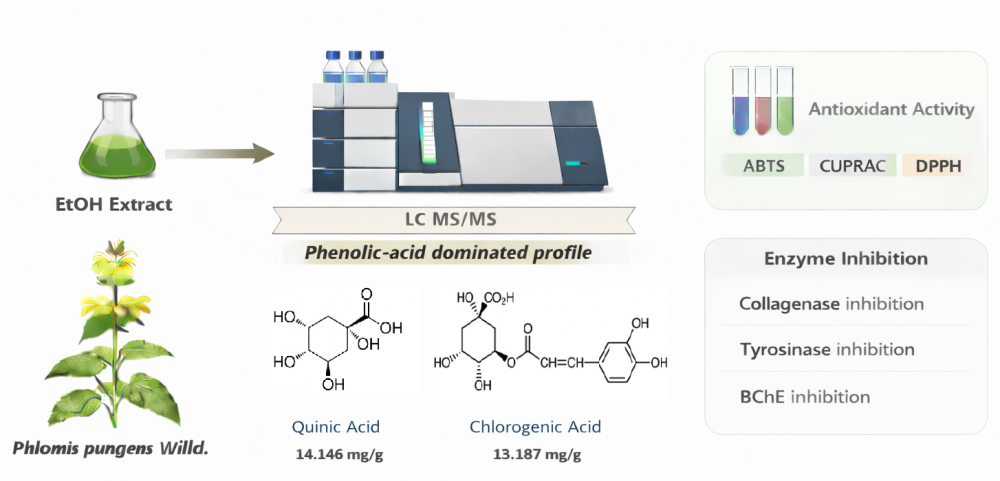
Phlomis pungens Willd. (syn. Phlomis herba-venti subsp. pungens (Willd.) Maire ex DeFilipps) is a medicinal and aromatic species that has been studied mainly for its essential oil composition, whereas its non-volatile phytochemical constituents have remained largely unexplored. In this study, the ethanolic extract of P. pungens was systematically characterized for the first time using a targeted LC–MS/MS method. In parallel, total phenolic and flavonoid contents, antioxidant potential, and multi-enzyme inhibitory activities were evaluated. LC–MS/MS profiling revealed a phenolic acid–rich composition, with quinic acid (14.146 mg/g extract) and chlorogenic acid (13.187 mg/g) identified as the predominant compounds.These were followed by cynaroside (1.541 mg/g), vanillic acid (1.453 mg/g), and cosmosiin (0.830 mg/g). The extract demonstrated considerable levels of total phenolics and flavonoids (37.18 ± 1.97 μg PEs/mg and 18.19 ± 0.90 μg QEs/mg extract, respectively). Antioxidant evaluation indicated moderate activity, particularly in the ABTS (IC50 = 32.05 ± 0.28 μg/mL) and CUPRAC (A0.5 = 20.83 ± 0.37 μg/mL) assays. Enzyme inhibition studies revealed selective inhibitory effects against collagenase (24.73 ± 0.32%), tyrosinase (15.04 ± 0.11%), and butyrylcholinesterase (13.62 ± 0.12%) at 50 μg/mL, while no inhibition was detected toward acetylcholinesterase, urease, or elastase.These findings suggest that the biological activities of P. pungens may be associated with its non-volatile phenolic constituents.This work provides novel phytochemical data and highlights the potential of the species for future pharmaceutical and dermocosmetic applications.
DOI http://doi.org/10.25135/rnp.2602.3755 Keywords Phlomis pungens LC–MS/MS phenolic compounds antioxidant activity enzyme inhibition medicinal plants DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.